The Disappearing Post: Are We Solving the Right Problem in Bone Conduction?
For decades, bone conduction hearing devices were defined by a small titanium post that protruded through the skin behind the ear. The abutment was visible, sometimes inconvenient, occasionally associated with skin complications—and yet mechanically, it worked remarkably well. By directly coupling the sound processor to the skull, percutaneous bone conduction systems provided efficient transmission of vibration to the cochlea and a predictable pathway for restoring access to sound (Håkansson et al., 1985; Wazen et al., 2010).
Today, that post is quietly disappearing.
In its place are magnet-based systems and fully implanted transducers designed to move the connection beneath the skin. The field has largely welcomed this shift. Active and passive transcutaneous devices offer potential advantages: reduced risk of skin infection, fewer maintenance requirements, and a more discreet appearance. For many patients, these are meaningful improvements (Dun et al., 2012; Sprinzl & Wolf-Magele, 2016).
But as bone conduction technology becomes increasingly sophisticated from a surgical and cosmetic perspective, an important question deserves renewed attention: are we solving the right problem?
The Physics of Transmission
The original percutaneous systems solved a fundamental engineering challenge—how to transmit vibration efficiently from the device to the skull. By eliminating the soft tissue barrier, the abutment created a direct mechanical pathway for sound energy.
When the connection moves beneath the skin, physics re-enters the equation.
Soft tissue attenuates vibration, particularly at higher frequencies. Passive transcutaneous systems must overcome this attenuation through stronger output and careful magnet coupling. Active systems attempt to bypass the soft tissue barrier by placing the transducer under the skin, closer to the skull, but introduce their own complexities in terms of surgical placement, device cost, and long-term management (Reinfeldt et al., 2015; Sprinzl & Wolf-Magele, 2016).
None of these trade-offs invalidate the benefits of transcutaneous approaches. For many patients, the advantages are substantial. But they remind us that eliminating the abutment does not eliminate the mechanical and acoustic constraints inherent in bone conduction transmission.
A Shift in What We Optimize
Historically, bone conduction development focused on acoustic performance: bandwidth, maximum power output, and reliable transmission to the cochlea.
More recently, much of the conversation has shifted toward surgical technique, cosmetic outcomes, and implant design. These are legitimate considerations. Patients deserve solutions that are comfortable, safe, and socially acceptable.
At the same time, the core purpose of any hearing device remains unchanged: restoring meaningful access to sound.
When the discussion centers primarily on what is visible—or invisible—it becomes easy to lose sight of the mechanical and acoustic performance that ultimately determines whether communication improves.
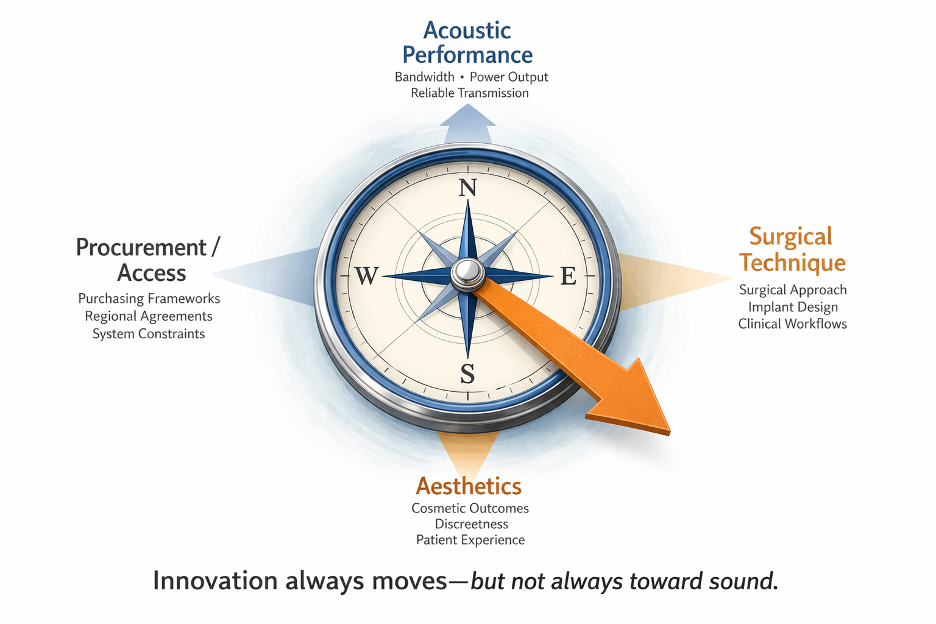
Image generated using OpenAI image-generation tools (DALL·E) based on author-defined conceptual input.
The Growing Complexity of Choice
Another consequence of innovation is the rapid expansion of device types. What was once a relatively straightforward category now includes percutaneous systems, passive transcutaneous implants, active transcutaneous implants, adhesive devices, and non-surgical headband solutions.
Each option carries different trade-offs in output capability, candidacy, surgical complexity, and cost. For clinicians, this growing menu of technologies can make decision-making less clear rather than more (Dun et al., 2012).
In principle, a broader set of options should empower clinicians and patients to find the best match for individual needs. In practice, however, access to these options is not always determined solely by clinical factors.
When Access Shapes Choice
In many jurisdictions, bone conduction devices are not simply selected based on patient characteristics or audiological outcomes. Instead, purchasing frameworks, hospital procurement systems, and regional agreements with manufacturers can determine which devices are available to clinicians and surgeons.
These arrangements are often designed to manage cost, simplify logistics, or support institutional partnerships. Yet they can have unintended consequences. When access to certain technologies is restricted at the jurisdictional level, clinical decision-making becomes intertwined with supply structures that may not always align with individual patient needs.
The result is a subtle but important shift: the range of solutions discussed in the clinic may reflect what can be purchased or implanted locally, rather than the full landscape of technologies available in the field.
This reality is not unique to bone conduction, but the relatively small and specialized nature of the market can amplify its effects.
Looking Ahead
None of this suggests that the transition toward transcutaneous systems is misguided. Innovation has produced devices that are more comfortable, more discreet, and increasingly sophisticated. Many patients benefit from these developments.
At the same time, the evolution of bone conduction technology invites reflection on where the next advances should occur.
Future progress may depend less on hiding hardware and more on addressing long-standing audiological challenges: improving maximum power output, extending high-frequency transmission, refining verification methods, and developing more precise fitting strategies. These are areas where acoustic science and clinical audiology remain central. Indeed, advances in prescription methods and verification approaches for bone conduction devices have already demonstrated that optimizing the acoustic fit can meaningfully influence patient outcomes (Hodgetts & Scollie, 2017).
The titanium post was never elegant. It was visible, imperfect, and occasionally troublesome. But acoustically, it represented a remarkably efficient solution to the problem of transmitting sound to the cochlea.
As the field continues to innovate, the challenge is not simply to make the post disappear. It is to ensure that, in doing so, we do not lose sight of the sound.
References
- Dun, C. A., Faber, H. T., de Wolf, M. J., Cremers, C. W., & Hol, M. K. (2012). An overview of different systems: The bone-anchored hearing aid. Advances in Oto-Rhino-Laryngology, 71, 22–31. https://doi.org/10.1159/000334144
- Håkansson, B., Tjellström, A., Rosenhall, U., & Carlsson, P. (1985). The bone-anchored hearing aid: Principal design and a psychoacoustical evaluation. Acta Otolaryngologica, 100(3–4), 229–239. https://doi.org/10.3109/00016488509121770
- Hodgetts, W. E., & Scollie, S. D. (2017). DSL prescriptive targets for bone conduction devices. International Journal of Audiology, 56(Suppl. 1), S48–S57. https://doi.org/10.1080/14992027.2016.1190531
- Reinfeldt, S., Håkansson, B., Taghavi, H., & Eeg-Olofsson, M. (2015). New developments in bone-conduction hearing implants: A review. Medical Devices: Evidence and Research, 8, 79–93. https://doi.org/10.2147/MDER.S39691
- Sprinzl, G. M., & Wolf-Magele, A. (2016). The Bonebridge bone conduction hearing implant: Indication criteria, surgery and a systematic review of the literature. Clinical Otolaryngology, 41(2), 131–143. https://doi.org/10.1111/coa.12484

