The Appeal of Signia’s Innovative Form Factors and Their Role in the Adoption of Hearing Aids

By: Andre Marcoux1, Brian Taylor2 and Daniel Olesen3
1 WS Audiology Canada Inc., Burlington, ON, Canada
2 Signia Inc., Iselin, NJ, U.S.A.
3 WS Audiology A/S, Lynge, Denmark
A previous article demonstrated how innovative product design decreases the stigmatizing effects of hearing aids for certain individuals who associate hearing loss as a negative societal attribute (Marcoux, Dostaler & Olesen, 2022). Using survey data, the authors discussed how product design, most notably Signia Active, a hearing aid developed to resemble the mainstream wireless earbud, could increase acceptance and permit individuals with hearing loss to obtain the necessary and timely help they need to live rich and active lives.
This article explores how stigma-reducing design concepts applied to hearing aids can promote increased their rate of adoption. Compared to many of its competitors, Signia offers three innovative form factors beyond the traditional portfolio of receiver-in-the-canal (RIC), behind-the-ear (BTE) and custom in-the-ear (ITE) devices. Signia’s innovative form factor portfolio consists of 1) Silk, an instant-fit, completely-in-the-canal (CIC), 2) Styletto, a slim RIC, and 3) Active, an instant-fit, in-the-canal (ITC). Each of the three form factors within the innovative portfolio addresses stigma by using a unique aspect of product design.
Signia Silk uses concealment, the most common product design strategy to create a solution for those with self-stigma. However, unlike other small devices which can be concealed in the ear canal, Silk has an outer exchangeable soft sleeve which follows the normal curvature of the ear canal and provides a comfortable instant-fit without the need for a custom tip. Since 2016, Silk has been the only prescription, instant-fit CIC on the market. As mentioned in Marcoux, Dostaler, & Olesen (2022): “While concealment is an effective design strategy in reducing the stigmatizing potential of hearing aids, the resulting miniaturization also presents its challenges and trade-offs. Smaller devices may not allow for larger batteries or receivers to provide the necessary power to correct more severe levels of hearing loss. Also, smaller devices may not have sufficient space to accommodate all the necessary sensors, circuits, and antennas to provide the same level of functionality the consumer has come to expect from slightly larger devices. For example, an invisible device which fits deeply into the ear canal may not have a sufficiently large footprint to accommodate an antenna for Bluetooth communication nor have sufficient line-of-sight for adequate reception and transmission”.
For its part, Signia Styletto addresses stigma by considering device de-identification in its product design. As stated in Marcoux, Dostaler & Olesen (2022): “ The purpose of device de-identification is to remove and modify the most identifiable features from the device as a means of decreasing its visibility and confusing any observer”. Also, “By removing the volume wheels and switches, as well as redesigning the housing of the hearing aid from the traditional oblong shape to one which is streamlined and elongated, the Styletto does not have the appearance of a typical behind-the-ear hearing aid”.
Finally, with Signia Active, the concept of device normalization was applied. The process of normalization is to design the stigmatizing device to become indiscernible from a non-stigmatizing target device (Marcoux, Dostaler & Olesen, 2022). Wireless earbuds have become increasingly popular for streaming music and calls from a user’s smartphone without the hassle of wires or large over-the-ear headphones. When designing hearing aids to resemble mainstream wireless earbuds, individuals who are concerned about the stigmatizing effects of traditional hearing aids are provided with an alternative option for correcting their hearing loss.
To validate whether these unique form factors of the innovative portfolio provide additional appeal to individuals with hearing loss, a study was done in which 384 Canadian respondents were surveyed. The research design, as well as a first subset of data, was first described in Marcoux, Dostaler & Olesen (2022). To briefly summarize the study design, respondents were recruited to complete an online questionnaire and were required to report hearing loss, as defined by a self-reported hearing loss questionnaire (Kochkin and Bentler 2010), hearing aid ownership, hearing loss diagnosis, previous interaction with hearing care professionals, as well as their gender and age. In this article, we report on a second subset of data consisting of respondents’ ratings of perceived interest, perceived effectiveness to solve a problem, and intent to trial and purchase hearing aids.
Respondents were first presented with an image corresponding to the left panel shown in Figure 1, where they viewed Signia RIC, BTE and ITC devices. They were told that a hearing center offers these three hearing aids and that all three meet the requirements of their hearing loss. They were then asked, if they would consider purchasing one of the three devices, or none of them. In a second round, each respondent again saw the same Signia RIC, BTE and ITC along with Styletto, Silk, and Active as shown in the right panel of Figure 1. The same question was asked, and again the respondents answered whether they would consider purchasing one of the devices or none of them.


We hypothesize that the addition of unique form factors increases the adoption of hearing aids. To test this hypothesis, we compared the number of participants who choose a hearing aid, versus choosing none, before and after the addition of the three new form factors to the offering. A non-parametric related-samples McNemar Change Test was performed. Results indicate a significant effect of the new form factors on reducing the number of non-buyers (X2(1)=2.118, p. <05). A non-buyer percentage of 8.9% was measured when only the traditional form factors were made available. With the addition of Styletto, Silk and Active, however, the percentage of non-buyers decreased to 7.0%, resulting in a total decrease of 20.6% in the number of non-buyers. It should be noted that the buyer rate in this study is greater that what is observed in the general population. This can be explained by the fact that 36% of respondents were current hearing aid wearers as well as the fact that common objections related to cost do not factor into their decision-making process in the study environment.
As this analysis does not confirm whether the share of non-buyers decreased as a result of simply having a greater number of options or because of the additional form factors themselves, other measures were also performed to determine which of the innovative form factors had an effect on the intent to trial or purchase the device, as well as their perceived uniqueness and effectiveness. Five questions were proposed to half of the respondents (n=196) which were randomly selected to evaluate the Signia ITC, RIC and BTE, while the other half (n=188) were asked to evaluate the Styletto, Silk and Active on a scale of 1 to 5 as shown in Table 1.
| Q1 What do you think about this hearing aid? | ||||
| I really do not like it | I do not like it | I like it somewhat | I like it | I really like it |
| Q2 How would you rate this hearing aid in terms of being new and different from other hearing aids currently available? | ||||
| Not at all new and different | Slightly new and different | Somewhat new and different | Very new and different | Extremely new and different |
| Q3 Which statement below best describes how well this hearing aid would solve a problem or fulfill a need for you? | ||||
| Definitely would not | Probably would not | Might or might not | Probably would | Definitely would |
| Q4 How much would you be interested in trying this hearing aid? | ||||
| Definitely would not like to try | Probably would not like to try | Might or might not like to try | Probably would like to try | Definitely would like to try |
| Q5 How much would you consider buying this hearing aid? | ||||
| Definitely would not buy | Probably would not buy | Might or might not buy | Probably would buy | Definitely would buy |
Table 1: Questions and answer scales for rating of each hearing aid.
When asked to evaluate a specific hearing aid, three views of the device were shown: a product picture, a side-view of the hearing aid on a person’s ear and a rear-view of the hearing aid on the ear of that same person, as illustrated in Figure 2.
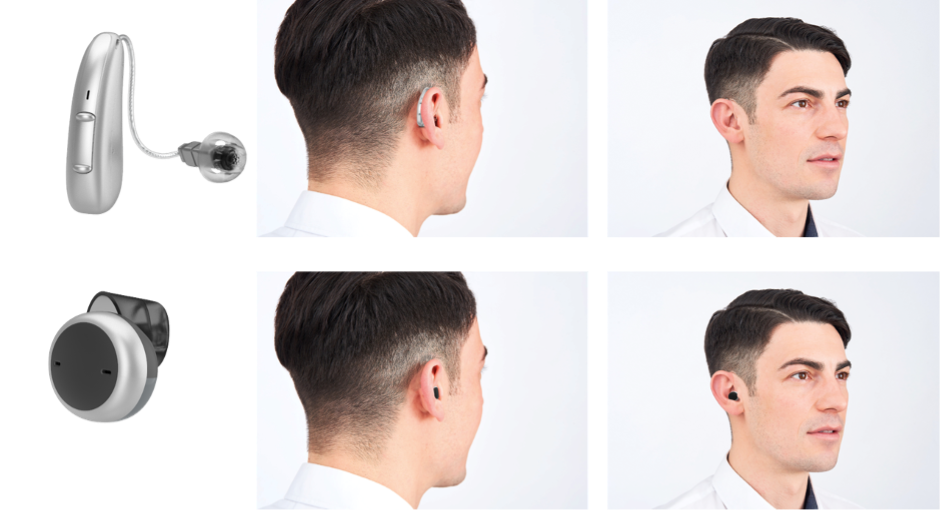
Figure 2. Device image as well as side and rear views of the device on a user’s ear used for rating. As examples, shown are Signia Pure Charge&Go in the top row and Signia Active in the bottom row.
Mean ratings from Group 1, which evaluated traditional form factors are labeled “traditional,” and Group 2, which evaluated innovative form factors are labeled “innovative” are reported in Table 2. Statistical analyses were performed to indicate significant differences in ratings between the three form factors within the assigned group. Significance (p<.05) from 2-sided t-tests are also indicated in Table 2 as superscript. As an example, in the traditional group, ITC is assigned as item 1, BTE as item 2 and RIC as item 3. For Question 1, ITC has a mean rating of 3,73 along with superscripted digits 2 and 3 in superscript, which indicates significantly different ratings than items 2 (BTE) and 3 (RIC). As indicated in Table 2, the ITC was significantly rated more favorably for the “traditional” form factor on all questions, while the Silk was significantly rated more favorably for the “innovative” form factors in terms of being new and different as well as on interest to trial and purchase. Interestingly, respondents in both groups rated Silk and the custom ITC as equally able to solve a problem and fulfill a need despite their small sizes. This may indicate that potential hearing aid wearers do not immediately realize that more feature-rich devices may be inherently larger, or that their need is linked to the desire to have a more concealable device. The greater ratings of attractiveness and effectiveness for the smaller devices also translate into a greater willingness to try and buy these devices.
| Group 1 (n=196) “Traditional” | Group 2 (n=188) “Innovative” | |||||
| ITC 1 | BTE 2 | RIC 3 | Styletto 1 | Silk 2 | Active 3 | |
| Q1 What do you think about this hearing aid? | 3,73 2,3 | 2,93 1 | 2,92 1 | 3,51 3 | 3,60 3 | 3,09 1,2 |
| Q2 How would you rate this hearing aid in terms of being new and different from other hearing aids currently available? | 3,30 2,3 | 2,13 1 | 2,10 1 | 2,76 2,3 | 3,43 1,3 | 3,25 1,2 |
| Q3 Which statement below best describes how well this hearing aid would solve a problem or fulfill a need for you? | 3,65 2,3 | 3,03 1 | 2,99 1 | 3,49 | 3,62 3 | 3,30 2 |
| Q4 How much would you be interested in trying this hearing aid? | 3,66 2,3 | 2,89 1 | 2,93 1 | 3,52 2,3 | 3,70 1,3 | 3,32 1,2 |
| Q5 How much would you consider buying this hearing aid? | 3,23 2,3 | 2,70 1 | 2,72 1 | 3,15 2,3 | 3,38 1,3 | 2,94 1,2 |
Table 2: Mean ratings for half of participant group rating ITC/Motion/Pure and the other half of the participant group rating Styletto/Silk/Active. Significant differences between form factors on each question are indicated in superscript.
Results of this survey indicate that individuals with hearing loss are significantly more likely to consider treatment when presented with a greater number of solutions; choices which include innovative form factors that differ from those traditionally available. Results also indicate that within the innovative portfolio, Silk provides added initial interest in trialling or purchasing a device. We believe this finding likely can be explained, at least in part, by stigma and the fact that stigmatized individuals with hearing loss tend to gravitate towards smaller, more concealable devices.
Additionally, we observed that among the devices worn behind the ear, Styletto was rated much more favorably that the traditional RIC or BTE. While Styletto was rated by Group 2 and cannot be directly compared to ratings provided by Group 1, which rated RIC and BTE devices, the large difference in ratings between Styletto and the RIC and BTE is noteworthy.
It is also noteworthy that all devices within the innovative portfolio were rated much more similarly, whereas there is a marked difference in ratings within the traditional portfolio where the RIC and BTE are rated much lower on all questions in comparison the custom ITC. This could indicate that all devices within the innovative portfolio have some appeal to persons with hearing loss as the innovative form factors provide different elements of product design which lead to greater consideration and adoption.
While these results indicate patterns of preference for small, innovative devices, they do not provide respondents’ ongoing ratings to each of these form factors once performance specification and audiological features are made known. Other studies are required to determine the contribution of product design, stigma, as well as other factors, responsible for the postponement of care.
We acknowledge the conundrum of promoting miniature devices to generate interest among persons with hearing loss as an approach to seeking prompt treatment. On one hand, promoting smaller devices will appeal to individuals with stigma and encourage them to seek the help they need, sooner. On the other hand, this same approach may exacerbate stigma by suggesting to hide their disability. Further complicating the promotion of smaller devices, most hearing care professionals are fully aware that they are not always suitable to meet the auditory and lifestyle requirements of some individuals because of their lower maximum output or limited number of features. While this conundrum will persist, and opinions will continue to differ, there is evidence that positive outcomes with hearing aids are linked to processes which promote the empowerment of the wearer (Gotowiek et al., 2021). In turn, an important element of empowerment is the individual’s attainment of knowledge about hearing loss and hearing aids. This requires a commitment from the professional to meet people “where they are” along with all their current notions and opinions, including those which pertain for a preference for smaller devices, to foster a dialogue leading to an effective and empowering level of care.
In a recent survey of more than 1200 middle-aged and older Americans, Carlson et al. (2022), reported limited knowledge and low levels of awareness on several questions related to hearing loss. While not studied directly, their results also suggest a lack of knowledge about treatment options, including the use of hearing aids and the pros and cons related to style, size, performance level, etc. In essence, while our survey indicated a strong appeal for concealable devices, it also represents an opportunity to create a discussion with potential wearers that speeds the journey toward earlier treatment and creates a path leading to greater empowerment and optimal outcomes.
A recent report indicates 85% of adults in the United States who self-report “hearing trouble” fail to seek treatment for their condition (Humes, 2023). Further, about one-third of adults with hearing loss complain that hearing aids are “too expensive” or that they lack insurance coverage (Carr, 2020). As over-the-counter (OTC) devices and the presence of big-box retailers have gained traction, out of pocket costs for hearing aids have trended downward. Yet with lower priced options, other adoption barriers, including stigma remain largely unchanged. According to an analysis by Williams (2021) of MarkeTrak 10 data, 21% of hearing aid non-owners cited stigma as a barrier to pursuing hearing aids. These respondents protested that hearing aids were “unattractive” or that they were “too young” to wear them. According to that same analysis by Williams (2021), 30% of hearing aid non-owners cited denial and indifference as other key barriers to acquiring hearing aids. We posit whether the underpinnings of denial and indifference could be attributable to stigma.
Many people who claim they “can hear well enough” or “have other priorities,” often delay acquiring hearing aids for several years. And, when they finally do begin wearing them, they experience suboptimal outcomes. Our results, presented here, suggest providing these help seeking individuals – whether they are in denial or believe hearing loss is a stigmatizing condition - with a greater choice of form factors which may be an effective clinical approach that significantly decreases the number of non-buyers. While patient outcomes hinge on the use of clinical best-practices, providing a broader choice of form factors promotes earlier interest in treatment of hearing loss, fosters deeper knowledge of hearing aids, and encourages timelier adoption of care from a hearing care professional.
References
- Carlson, M.L., Nassiri, A.M., Marinelli, J.P., Lohse, C.M., Sydlowski, S.A. (2022). Awareness, perceptions, and literacy surrounding hearing loss and hearing rehabilitation among the adult population in the United States. Otolology and Neurotololy, 43(3), 323-330.
- Carr, K. (2020). 20Q: Consumer insights on hearing aids, PSAPs, OTC devices, and more from MarkeTrak 10. AudiologyOnline, Article 26648.
- Gotowiec, S., Larsson, J., Incerti, P., Young, T., Smeds, C., Wolters, F., Herrlin P., & Ferguson, M. (2021): Understanding patient empowerment along the hearing health journey, International Journal of Audiology, 61(2), 148-158.
- Humes L. E. (2023). U.S. Population Data on Hearing Loss, Trouble Hearing, and Hearing-Device Use in Adults: National Health and Nutrition Examination Survey, 2011-12, 2015-16, and 2017-20. Trends in hearing, 27, 23312165231160978.
- Kochkin, S., & Bentler, R. (2010). The validity and reliability of the BHI Quick Hearing Check. Hearing Review, 17(12), 12-28.
- Marcoux, A., Dostaler, M., & Olesen, D. (2022). Signia Active: The beginning of an era of hearing without stigma. Canadian Audiologist, 9(1).
- Williams, N. (2021). Addressing hearing loss stigma within the OTC context. Auditory Insight Research Note, Q3. Downloaded at: https://auditoryinsight.com/wp-content/uploads/securepdfs/2021/11/Auditory_Insight_Stigma_AntiSmoking_Analog_Q3_2021.pdf

