Other People’s Ideas
Calvin Staples, MSc, will be selecting some of the more interesting blogs from HearingHealthMatters.org which now has almost a half a million hits each month. This blog is the most well read and best respected in the hearing health care industry and Calvin will make a regular selection of some of the best entries for his column, Other People’s Ideas.
The monthly blog submission is always something I enjoy. The folks at hearinghealthmatters.org do a wonderful job providing clinicians with some insight regarding the advances seen in audiology. The pharmacological updates in audiology seem to occur weekly and to me are easily the most fascinating. If or when any of these updates come to clinical practice I have no idea, but I think as clinicians it is imperative we are aware of some of these developments so that we can craft out our role as we move forward and best serve our patients. Happy reading!
Anti-Stroke Drug Holds Promise In Fight Against Ear Infections, Say Researchers
Originally posted at HHTM On June 9, 2015. Reprinted with permission

Kensei Komatsu, PhD, study co-author
ATLANTA– Researchers from Georgia State University and the University of Rochester may have found a non-antibiotic alternative to treating middle ear infections.
In a study published this month in the Journal of Immunology, the anti-stroke drug vinpocetine showed the ability to improve bacterial clearance, suppress mucus overproduction, and reduced levels of hearing loss in treated mice.
Middle ear infections (otitis media) are one of the most common reasons why children visit the pediatrician–over 24 million physician visits per year in the US alone. Billions of dollars are spent annually due to the direct and indirect costs involved with treating otitis media.[1]
The Most Common Cause of Childhood Hearing Loss
Conductive hearing loss due to otitis media is the most common cause of hearing loss in children. While otitis media is usually not very serious, in some cases it can become chronic, and effective treatment can be difficult to achieve.
Surgery to place ventilation tubes. Image courtesy Wikimedia Commons, Erica Mater
In persistent and recurrent cases of otitis media in childhood, the fluctuating mild to moderate levels of hearing loss have been found to result in speech and language delays. After repeated infections, doctors may recommend surgical placement of ventilation tubes into the eardrums to help improve fluid drainage and minimize the risk of future infections.
According to the American Academy of Otolaryngology, more than half a million ear tube surgeries are performed on children every year, which makes it the most common childhood surgery performed with anesthesia.
Encouraging Results
Antibiotics have been the primary method of treating otitis media for many years, since an effective non-antibiotic treatment has not yet been found. However, the increasing use of antibiotics on both humans and animals has led to the emergence of bacteria that are resistant to antibiotics. As concern grows that some of the antibiotics that are used today may become ineffective, doctors are likely to welcome the development of non-antibiotic treatments.
In the current study, a topical application of vinpocetine was used against the bacteria Streptococcus pneumoniae—the primary cause of inflammation and over-production of mucus seen in otitis media. The treated animals were found to have suppressed inflammation, reduced mucus production, and improved hearing.

Periwinkle plant
Vinpocetine is a man-made synthetic chemical resembling the extract derived from the periwinkle plant Vinca minor. Marketed under the trade name Cavinton since 1978, It has been used around the world to treat cerebrovascular disorders. Although it is not currently approved in the United States for pharmaceutical use, vinpocetine can be purchased as a dietary supplement.
The researchers report that during the drug’s history:
“There have been no reports of significant side effects and toxicity related to vinpocetine treatment at therapeutic doses, and various clinical studies have demonstrated that vinpocetine is safe for long-term use.”
Researchers believe that the drug could be re-purposed from its current usage to create an effective topical treatment agent for otitis media, which could be applied directly to the affected area. They are hopeful that the findings will lead to the development of the first effective non-antibiotic drug treatment for millions of people that suffer from middle ear infections each year.
Source: Georgia State University
Images courtesy wikipedia, Georgia State University
Footnotes
- Gates GA (1996) Cost-effectiveness considerations in the otitis media treatment, Otolaryngol Head Neck Surg 114:525
Disease Modifying Drugs for Hearing Loss
Originally posted at HHTM on February 4, 2014. Reprinted with permission.
Most audiologists know that the ability to hear relies upon small sensory cells or “hair cells” in the inner ear. Many types of commonly acquired hearing loss are the result of damage or death to these delicate hair cells, including noise-induced hearing loss, presbycusis (age-related hearing loss), and ototoxic hearing loss (hearing loss that occurs after a patient is exposed to certain life-saving but toxic medications).
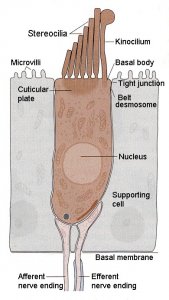
More than 90% of hearing loss occurs when either the hair cells or auditory nerve cells are destroyed. This starts affecting some individuals from birth and about one-third of the world’s population over age 65 and about half of those 75 or older.
Named for the microscopic hair-like extensions called “stereocilia” projecting from their tops in bundles, these “hair bundles” (right) convert sound vibrations into electrical impulses that travel to the brain via CN VIII, the auditory or hearing nerve.
Once damaged in mammals, these hair cells are gone forever, leaving humans hearing impaired. Until the last few years, scientists believed that hair cells in mammals could never be replaced if they were injured or destroyed. In the late 1980s, researchers discovered that hair cells could be regenerated by the Japanese quail. This discovery led to hopes that hair cells could also be reproduced by humans when given the correct combination of ingredients. Its now been over 25 years since the discovery that birds can regenerate hair cells (Corwin & Cotanche, 1988;Ryals & Rubel, 1988). While the research looks promising, (the Hearing Pill, notwithstanding) there is still no approved disease modifying drugs for sensori-neural hearing loss. Through gene therapy and stem cell research, however, scientists have been able to grow new hair cells in laboratory animals, in some cases restoring some hearing to deafened mammals.

As with other important scientific discoveries such as penicillin, the discovery that chickens can regenerate their own hearing occurred almost by accident. While studying how drugs that cause hearing damage affect the tiny sensory cells in the ear, Hearing Health Foundation funded scientists needed to permanently damage a chicken’s hair cells. (Click on the Chicks for a video.) For 10 days, research assistants administered a common antibiotic known to cause hearing loss to laboratory chickens. On day 11 many of the hair cells were gone and a few days later, even more had disappeared. Surprisingly, when the scientists looked three weeks later, almost all the hair cells had returned. They didn’t believe these results so they did the experiment again and again. Sure enough, chickens can naturally regenerate their inner ear hair cells, restoring their hearing after damage. Now we know this ability is true of all types of vertebrate animals except mammals, including us.
Experiments with mice are also proving to be enlightening. In these studies, researchers applied a drug to the cochlea of deaf mice. The drug had been selected for its ability to generate hair cells when added to stem cells isolated from the ear. It acted by inhibiting an enzyme called gamma-secretase that activates a number of cellular pathways. The drug applied to the cochlea inhibited a signal generated by a protein called Notch on the surface of cells that surround hair cells. These supporting cells turned into new hair cells upon treatment with the drug. Replacing hair cells improved hearing in the mice, and the improved hearing could be traced to the areas in which supporting cells had become new hair cells.
“The missing hair cells had been replaced by new hair where the hair cells were replaced,” said Dr. Albert Edge of Massachusetts Eye and Ear Infirmary and Harvard Medical School.
Thus, replacing hair cells improved hearing in the mice, and the improved hearing could be traced to the areas in which supporting cells had become new hair cells. According to Dr. Edge, this publication of hair cell regeneration in Neuron last year was the first demonstration of hair cell regeneration in an adult mammal.

“We’re excited about these results because they are a step forward in the biology of regeneration and prove that mammalian hair cells have the capacity to regenerate,” Dr. Edge said. “With more research, we think that regeneration of hair cells opens the door to potential therapeutic applications in deafness.”
The key to restoring hearing in humans is to regenerate cells deep within the inner ear. In fact, most types of hearing loss in humans results from damage to these cells, called hair cells. While humans cannot regenerate hair cells in the inner ear after they are damaged, chickens can. In fact, most animals other than mammals can regenerate these delicate cells, restoring their hearing spontaneously after damage. Our goal is to translate this to humans.
……and Then What?
A few years ago, Ryals (2009) indicated that once these hair cell regeneration options were available for hearing impaired patients, clinical audiologists will be faced with the responsibility of translating these basic science findings into potential patient application. She suggested that the possibility of curing hearing loss might raise important questions, such as…..When will hair cell regeneration be a reality for my patients? What will be the measures of candidacy? What will the impact of hair cell regeneration be in my patients who use or are candidates for hearing aids or other amplification devices? Will hearing aids or cochlear implants continue to be needed in the face of hair cell regeneration?
We are now much closer to having to answer these questions than ever before!
Scientists Develop A Way To Induce Temporary Hearing Loss In Human Subjects
Originally posted on February 6, 2013. Reprinted with permission
GAINESVILLE, FL—Hearing scientists have learned much of value about hearing, hearing loss, and its causes and treatments by conducting research experiments on laboratory animals. However, no matter how promising a procedure or drug seems to be in animal studies, at some point before a new treatment is made available to the public, researchers must find out if what worked on mice or chinchillas is also effective for humans.
Designing human clinical trials is complicated by issues of safety and ethics that do not arise with research subjects of other species. For example, prototype drugs have been developed that prevent a temporary noise-induced hearing loss in lab animals from becoming permanent. But it has been difficult for researchers to determine if such drugs will do the same for humans. That is largely because they lacked an effective method of causing a temporary, reversible hearing threshold shift in human subjects that they could be certain would not become a permanent hearing loss.
Recently, however, a group of scientists reported in Ear and Hearing that they had developed a real-world digital music exposure that reliably induces temporary threshold shift (TTS) in normal-hearing human subjects.
The lead investigator, Colleen Le Prell, PhD, an associate professor in the Department of Speech, Language, and Hearing Sciences at the University of Florida, and her colleagues enlisted 33 subjects for the studies, which measured the effects of digital music player use on hearing. Subjects selected either rock or pop music, which was presented to them for four hours on a digital music player via headphones. The music was presented at three different levels: 93 to 95 dBA to 10 subjects, 98 to 100 dBA to 11 subjects, and 100 to 102 dBA to 12 subjects.
Audiograms and distortion product otoacoustic emissions (DPOAEs) were measured before the music exposure and then four times during the first 3-1/4 hours after exposure and again one day and one week after the test. Fifteen minutes after the music stopped, those who listened to the highest music levels had lost a small amount of hearing—six dB, on average. Their hearing returned to normal within three hours.
In their conclusion, Le Prell et al. state, “These data provide insight into the variability of TTS induced by music-player use in a healthy, normal-hearing, young adult population, with music playlist, level, and duration carefully controlled. These data confirm the likelihood of temporary changes in auditory function after digital music-player use.
“Such data are essential for the development of a human clinical trial protocol that provides a highly powered design for evaluating novel therapeutics in human clinical trials.
“Care must be taken to fully inform potential subjects in future TTS studies, including protective agent evaluations, that some noise exposures have resulted in neural degeneration in animal models, even when both audiometric thresholds and DPOAE levels returned to pre-exposure values.”
Le Prell’s group plan to use this testing model in two clinical trials of therapeutics designed to determine if noise-induced hearing loss can be prevented in humans.
Interviewed for the University of Florida News, Le Prell said, “We really want to find out what’s going to work and we want to make it possible for strategies that do work to get in the hands of the people who need them.”
In the same article, Jianxin Bao, an associate professor at Washington University School of Medicine in St. Louis and an expert on hearing, said, “Dr. Le Prell started with a unique idea to create a reversible noise-induced hearing loss and has established solid groundwork for this new model in the use of clinical drug testing.”
Dr. Bao, who was not involved in the study, added, “As for every new model, several unknown factors exist for this elegant experiment model, which requires further detailed studies.”
Testing Begins On Drug Treatment for Hearing Loss And Tinnitus
Orignally posted on June 11, 2013. Used with permission.

Charles Large
LONDON–Autifony Therapeutics, a British startup biotech company, haslaunched a Phase 1 trial of an experimental drug, AUT00063, that is designed to treat age-related hearing loss and tinnitus by targeting deficits in auditory processing in the brain that are believed to cause these problems.
Charles Large, PhD, co-founder and chief scientific officer of Autifony, noted, “We are delighted by the progress that has been made to bring AUT00063 into clinical development. Hearing loss and tinnitus affect the lives of a vast proportion of the population, yet there are no effective treatments available.” He added that AUT00063, a potassium channel modulator, has the potential to change that.”
The trial, which involves about 60 subjects, both young and old, will assess safety and dosing of AUT00063 in the treatment of hearing loss and tinnitus. The company expects to complete the trial by the first quarter of 2014.
Autifony, which was spun off from the pharmaceutical giant GlaxoSmithKline (GSK) in 2011, has received nearly $25 million in funding from companies, including Pfizer Venture Investments. It works closely with hearing research experts at University College London’s Ear Institute, Yale University, and other academic collaborators around the world.

