Mysteries of the Hearing Brain — What Can Rate Code Tell Us About Cochlear-Implant and Older Listeners?
In the last issue of Canadian Audiologist (March, 2020), I wrote about potential uses of real-world signals in clinical EEG testing. In this issue, I will describe the use of highly controlled, basic stimuli to investigate the difficulties experienced by older adults or cochlear implant (CI) users when trying to converse in a multi-talker environment. The use of basic stimuli may help to answer specific questions about auditory processing deficits that may be contributing to these difficulties. To better understand the nature of auditory processing deficits, it is useful to review how the auditory system encodes sound. Several types of neural coding are used to process sound. Rate code refers to the discharge rate of the neuron that codes frequency or intensity. Place code refers to frequency coding through tonotopicity in the cochlea or other structures in the auditory system. Phase-locking refers to frequency coding through neural firing that corresponds to the period of the stimulus (e.g., a 100-Hz tone would have peaks occurring every 10 ms – the stimulus period). Finally, the ensemble code is used to code information from populations of neurons regarding the frequency, timing, and intensity of the stimulus. The evaluation of the auditory system’s ability to accurately represent these codes may help to understand differences in speech understanding abilities in certain clinical populations, such aging listeners or cochlear implant (CI) users. The ability to detect changes in stimulus rate, for example, may help to explain aging deficits in fundamental frequency discrimination, affecting identification of a voice on the basis of gender.1 When a person finds oneself in an environment with multiple talkers, voice pitch (fundamental frequency) is an important cue for being able to focus on the target talker.2Therefore, impaired rate discrimination may affect an older person’s ability to understand speech in a cocktail party scenario.
CI listeners rely primarily on temporal cues (rate code) to discriminate fundamental frequency; however, their rate discrimination is generally limited to 300 Hz, whereas normal-hearing (NH) listeners can discriminate acoustic pulse trains up to 700-800 Hz.3However, these differences between CI and NH listeners may be due to aging rather than to hearing impairment or CI limitations. To better understand the nature of these differences, we recorded auditory steady-state responses (ASSRs) to 300-ms acoustic pulse trains with a 300-ms interstimulus interval at five different modulation rates (20, 40, 80, 200, and 400 Hz) in 15 younger and 15 older NH listeners.4 Spectral energy corresponding to each rate is displayed in Figure 1, and higher energy is depicted with hotter (redder) colors. It is apparent that both the younger and older listeners show neural energy corresponding to modulation rate up to 200 Hz. But, at 400 Hz, the older listeners’ responses drop off significantly while the younger listeners responses are maintained. This aging deficit specific to relatively higher frequencies is consistent with other ASSR studies evaluating aging effects.5,6
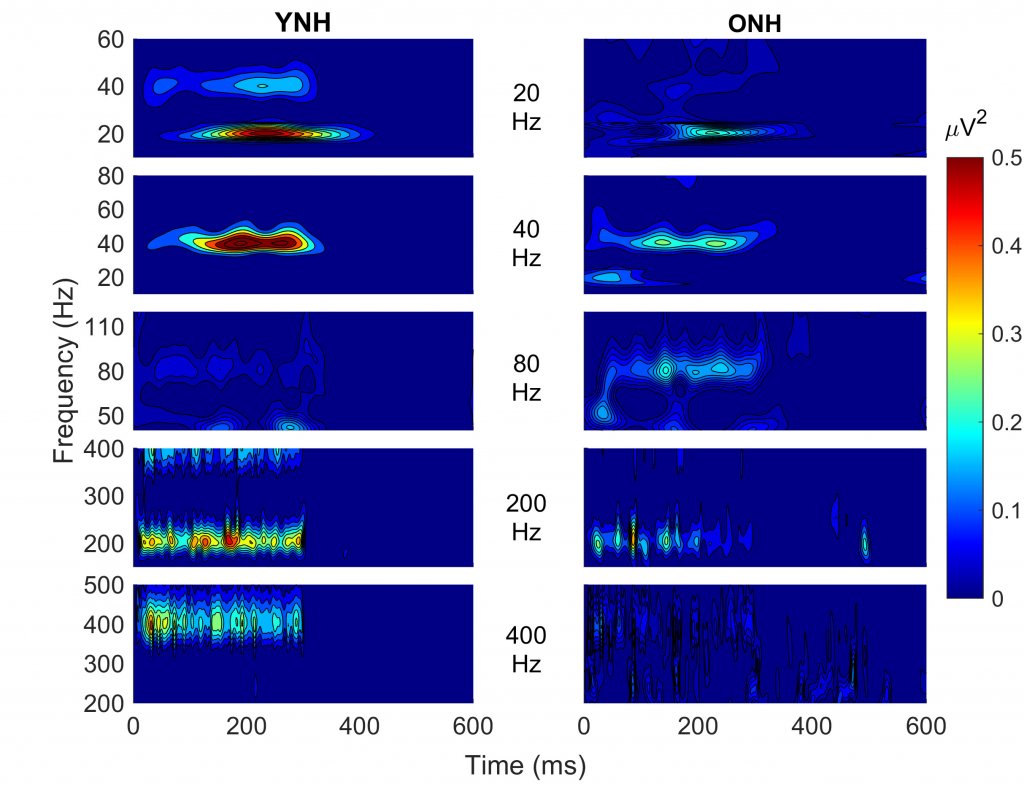
Figure 1. Spectral energy is displayed from auditory steady-state responses recorded to acoustic pulse trains presented at different modulation rates in young normal-hearing (YNH) and older normal-hearing (ONH) listeners. Higher energy is depicted with hotter (redder) colors. Both YNH and ONH listeners show spectral energy corresponding to the modulation rate up to 200 Hz, but at 400 Hz a marked decline is noted in the ONH listeners, but not in the YNH listeners.
Perceptual rate discrimination was also measured in these listeners, and performance was equivalent between the two age groups. When measuring behavioral performance, it is possible that other factors contribute to successful performance, such as listening effort and attention. In lab experiments or during clinic appointments, older adults tend to exert effort to achieve their best performance; however, this level of effort is not sustainable for long periods of time in their own environments. Therefore, objective measures of neural processing may provide a more sensitive measure of aging deficits.
Returning to older CI listeners, more work needs to be done to determine if their listening difficulties are due to age, duration of deafness, or other factors. It is hoped that a better understanding of these factors will lead to individualized treatment (e.g., CI mapping parameters) and better speech understanding outcomes.7
References
- Souza P, et al. Effects of age on F0 discrimination and intonation perception in simulated electric and electroacoustic hearing. Ear Hear 2011;32(1):75–83. https://doi.org/10.1097/AUD.0b013e3181eccfe9.
- Oxenham A.J. Pitch perception and auditory stream segregation: Implications for hearing loss and cochlear implants. Trends Amplif 2008;12(4):316–31. https://doi.org/10.1177/1084713808325881.
- Carlyon RP and Deeks JM. Limitations on rate discrimination. J Acoust Soc Ame 2002;112(3 Pt 1):1009–25. https://doi.org/10.1121/1.1496766.
- Gaskins C, et al. Effects of aging on perceptual and electrophysiological responses to acoustic pulse trains as a function of rate. J Speech Langugae Hear Res 2019;62(4s):1087–98. https://doi.org/10.1044/2018_jslhr-h-ascc7-18-0133.
- Grose JH, Mamo SK and Hall JW 3rd, Age effects in temporal envelope processing: speech unmasking and auditory steady state responses. Ear Hear 2009;30(5):568–75. https://doi.org/10.1097/AUD.0b013e3181ac128f.
- Leigh-Paffenroth ED and Fowler CG Amplitude-modulated auditory steady-state responses in younger and older listeners. J Am Acad Audiol 2006;17(8):582–97. https://doi.org/10.3766/jaaa.17.8.5.
- Shader MJ, et al, The effect of stimulation rate on speech understanding in older cochlear-implant users. Ear and Hearing 2019. Available at: https://doi.org/10.1097/AUD.0000000000000793.

