Mysteries of the Hearing Brain: Aging and Cochlear Implants
Years (actually decades) ago, I found it challenging to work with patients facing rapidly progressing sensorineural hearing loss. Before the advent of multi-channel cochlear implants (CIs) in ~1984, these patients had few options for maintaining the ability to communicate in a hearing world. Auditory training and lip reading strategies provided limited benefit, and patients found it difficult to learn sign language in older age. Although I have never been directly involved in cochlear implant fitting, my practice changed dramatically when cochlear implantation became a viable solution for many patients. My patients with progressive hearing loss no longer dreaded their audiology appointments but actually anticipated when their hearing losses would progress to the point of cochlear implant candidacy. I remember, in particular, one patient who was experiencing progressive vision and hearing loss associated with Usher’s syndrome. The joy that she communicated to me after being able to hear again through her cochlear implant was a highlight of my career.
The benefits of CIs can be amazing, even for our elderly patients. In my Introduction to Audiology class for undergraduates, I briefly introduce hearing aids and CIs, and I try to help them understand when CIs might be a better solution than hearing aids for severe hearing loss. I remember one final exam that included a case study question on this topic. The patient was a 55-year-old woman wearing hearing aids for many years but was no longer receiving adequate benefit because of decreased hearing thresholds and word recognition scores of ~ 20% in both ears. One of my students had written that she did not think this woman would benefit from a cochlear implant because she was “so old.” Seriously? I happened to be 55 years old when I taught the class, and I was not amused!
Fortunately, many elderly patients have experienced dramatically improved quality of life, even after being implanted in their 90s and older. But, during preimplant and postimplant counseling, it is important to recognize that older patients may not experience outcomes as high as those of younger cochlear implant recipients. Duration of deafness is the strongest predictor of outcomes, and older listeners tend to have longer durations of deafness. Aging itself also appears to be a factor in successful outcomes. Temporal processing, which decreases with age, is a strong factor in understanding speech in normal-hearing listeners, especially in challenging listening situations (Gordon-Salant and Fitzgibbons, 1993; Zhou et al., 2019). Aging also affects temporal processing in older CI listeners. For example, detecting small fluctuations in amplitude modulations is crucial to understanding speech, and aging reduces the ability to detect amplitude modulations in CI listeners (Shader et al., 2020).
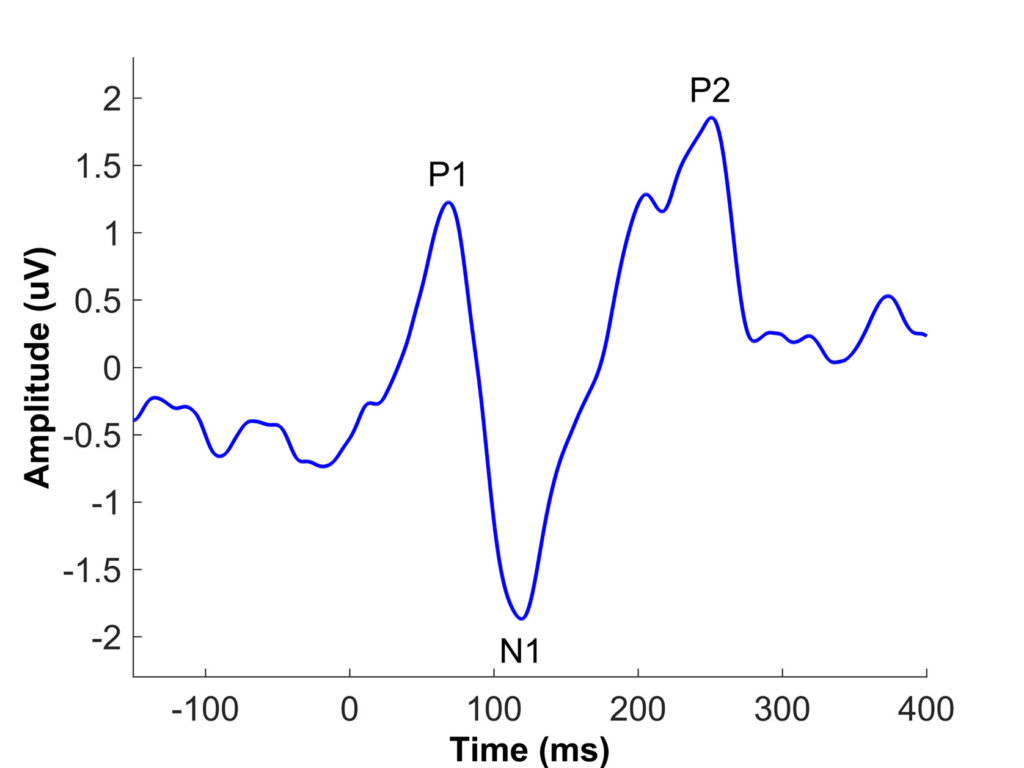
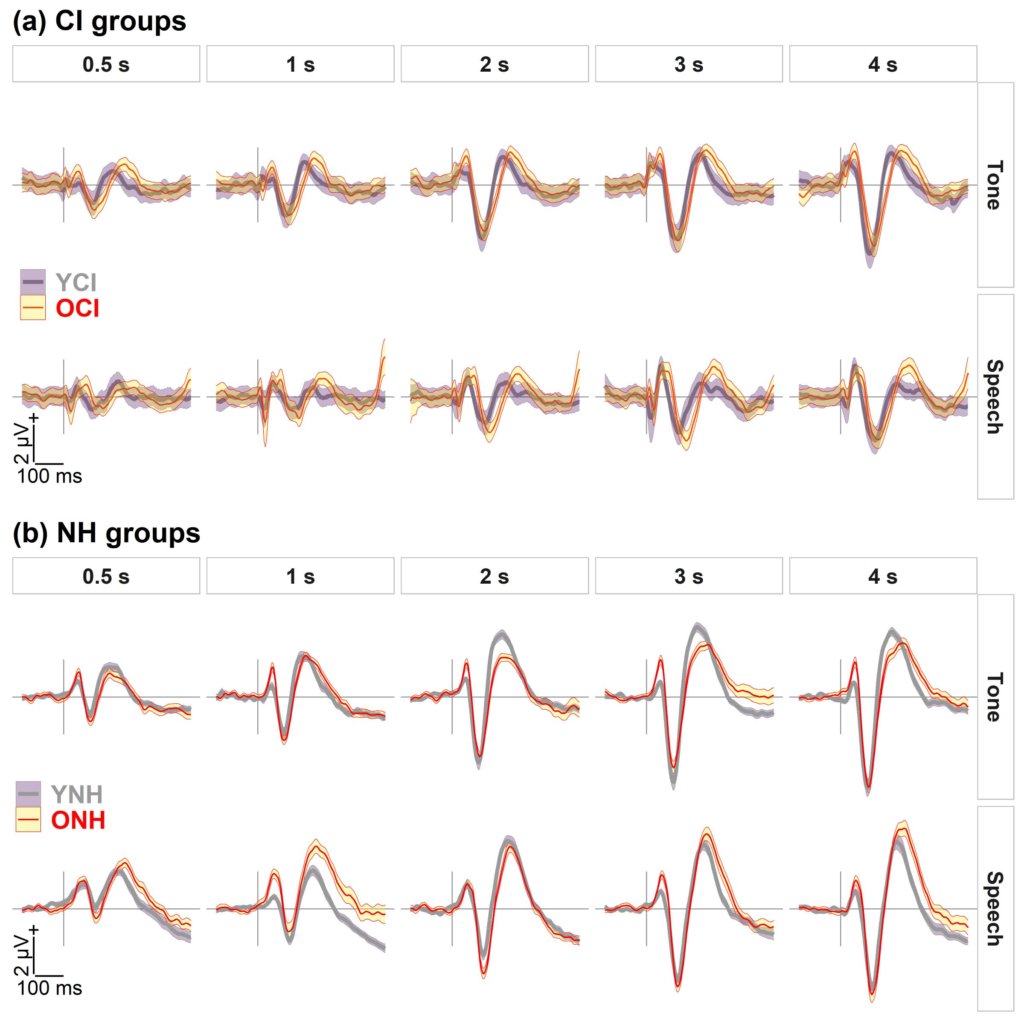
Xie et al. (2021) used cortical auditory-evoked potentials (CAEPs) to investigate potential neural sources of speech understanding deficits in older CI listeners. The primary CAEP peaks in adults include P1, N1, and P2 (see Figure 1). The latency, or the timing of these peaks, is considered an objective measure of auditory temporal processing, and previous aging studies have shown age-related latency delays of cortical peaks, especially N1 and P2 (Roque et al., 2019; Tremblay et al., 2004). Xie et al. (2021) recorded CAEPs to a 1000-Hz tone and a speech syllable /da/ presented at interstimulus intervals ranging from 0.5 to 4 s in middle-aged and older listeners with CIs and in age-matched listeners with normal hearing (NH). They found that older CI listeners had delayed latencies for the N1 and P2 peaks in responses to the /da/ syllable and delayed latencies for the P2 peak in response to the 1000-Hz tone compared to younger CI listeners (see Figure 2). The NH older listeners also had prolonged P2 latencies compared to the NH younger listeners replicating previous studies. In previous studies, a hearing loss confound has limited the interpretation of aging effects in NH listeners, because older listeners nearly always have worse hearing thresholds than younger listeners (Anderson and Karawani, 2020). The Xie et al. (2019) study is important because the cochlear implant bypasses the cochlea; therefore, the results support the idea that centrally based temporal processing deficits affect the older CI listeners. This reduced temporal processing may limit the older CI listener’s ability to understand speech, especially in noise or in other challenging listening situations. It is hoped that in the future, the CAEP can be used as an additional tool to evaluate temporal processing in CI listeners, so that they can be counseled regarding appropriate expectations for speech understanding ability.
References
- Anderson, S., Karawani, H. 2020. Objective evidence of temporal processing deficits in older adults. Hear. Res. 397, 108053, 10.1016/j.heares.2020.108053.
- Gordon-Salant, S., Fitzgibbons, P.J. 1993. Temporal factors and speech recognition performance in young and elderly listeners. J. Speech Hear. Res. 36, 1276-1285, http://dx.doi.org/10.1044/jshr.3606.1276.
- Roque, L., Gaskins, C., Gordon-Salant, S., Goupell, M.J., Anderson, S. 2019. Age effects on neural representation and perception of silence duration cues in speech. J. Speech Lang. Hear. Res. 62, 1099-1116, https://doi.org/10.1044/2018_jslhr-h-ascc7-18-0076.
- Shader, M.J., Gordon-Salant, S., Goupell, M.J. 2020. Impact of Aging and the Electrode-to-Neural Interface on Temporal Processing Ability in Cochlear-Implant Users: Amplitude-Modulation Detection Thresholds. Trends Hear 24, 2331216520936160, 10.1177/2331216520936160.
- Tremblay, K., Billings, C., Rohila, N. 2004. Speech evoked cortical potentials: Effects of age and stimulus presentation rate. J. Am. Acad. Audiol. 15, 226-237, https://doi.org/10.3766/jaaa.15.3.5.
- Xie, Z., Stakhovskaya, O., Goupell, M.J., Anderson, S. 2021. Aging Effects on Cortical Responses to Tones and Speech in Adult Cochlear-Implant Users. J. Assoc. Res. Otolaryngol. 22, 719-740, 10.1007/s10162-021-00804-4.
- Xie, Z., Gaskins, C.R., Shader, M.J., Gordon-Salant, S., Anderson, S., Goupell, M.J. 2019. Age-Related Temporal Processing Deficits in Word Segments in Adult Cochlear-Implant Users. Trends Hear. 23, 1-19, 10.1177/2331216519886688.
- Zhou, N., Mathews, J., Dong, L. 2019. Pulse-rate discrimination deficit in cochlear implant users: is the upper limit of pitch peripheral or central? Hear. Res. 371, 1-10, 10.1016/j.heares.2018.10.018.

